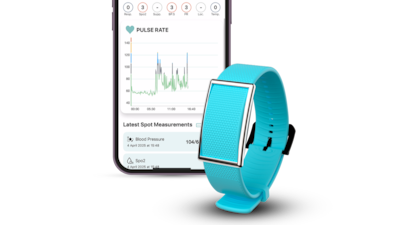
Jeremiah Robison is on his way to transform his passion project to help his daughter, who was diagnosed with cerebral palsy at 14 months of age, into a medical-grade device to help others with mobility issues.
The CEO of San Francisco-based Cionic and his team are developing a garment or “Neural Sleeve” that uses smart sensors...