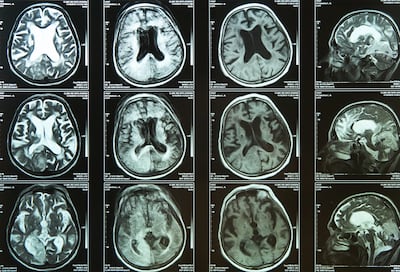
Nines recently became the first company to gain 510(k) clearance from the US Food and Drug Administration to market an algorithm-based software solution called NinesAI that allows radiologists to triage intracranial hemorrhage and mass effect on computed tomography (CT) scans. Mass effect, which is associated with such conditions as stroke and brain tumor, causes the surrounding areas of the brain to be compressed.
Nines’ co-founder and CEO David Stavens said it will make the technology available to existing and new customers who sign...
Read the full article – start your free trial today!
Join thousands of industry professionals who rely on Medtech Insight for daily insights
- Start your 7-day free trial
- Explore trusted news, analysis, and insights
- Access comprehensive global coverage
- Enjoy instant access – no credit card required
Already a subscriber?